DFT Analysis of the Gas Phase Interactions of Choline Chloride with Urea
Dan E. Diendere, Edouard Tapsoba, Françoise Diendere, Moussa R. Bougouma
American Journal of Chemistry, 2026, 16(1), pp. 1-20
DOI: 10.5923/j.chemistry.20261601.01
Open Access
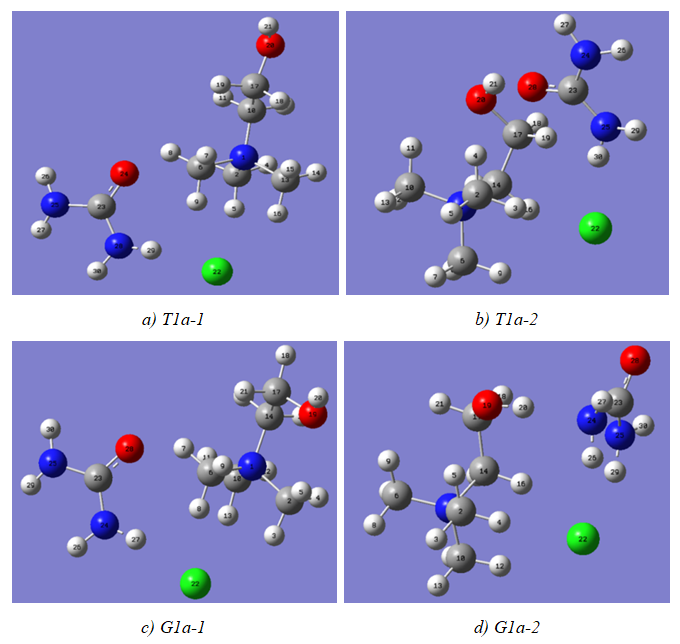
Choline chloride and urea interact to form a Deep Eutectic Solvent widely used as a green solvent in several applications such as electrochemistry, chemical substance separation and reactions. We searched for the equilibrium geometries, charges distributions of model compounds of their respective ...
Full-Text HTMLDownload Full-Text PDF
2250 Views 333 Downloads
Optimization of Surgical Treatment of Postoperative Ventral Hernias
Bakhriyev Bakhrom Lapasovich, Davlatov Salim Sulaymonovich, Rakhmanov Kosim Erdanovich, Shirinova Shakhinabonu Bobirovna
American Journal of Medicine and Medical Sciences, 2026, 16(1), pp. 5-9
DOI: 10.5923/j.ajmms.20261601.02
Open Access
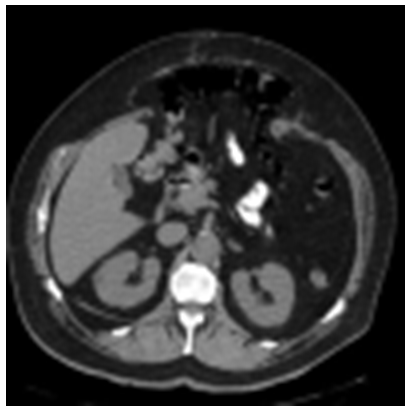
Introduction. Incisional ventral hernias (IVHs) represent a significant problem in surgical practice, and their incidence varies depending on various factors, including the type of surgery, patient age, gender, and the presence of comorbidities. ...
Full-Text HTMLDownload Full-Text PDF
406 Views 291 Downloads
Application of Chitosan and Egg White Based Organic Coating on Cotton as Water Resistant
Tanzeena Refat Tumpa, Sunchyen Barua, Tanzim Tabassum, Md. Wahidur Rahman, Md. Lutfor Rahman, Muhammad Tanjil, Riffat Islam
International Journal of Textile Science, 2025, 14(2), pp. 21-26
DOI: 10.5923/j.textile.20251402.01
Open Access
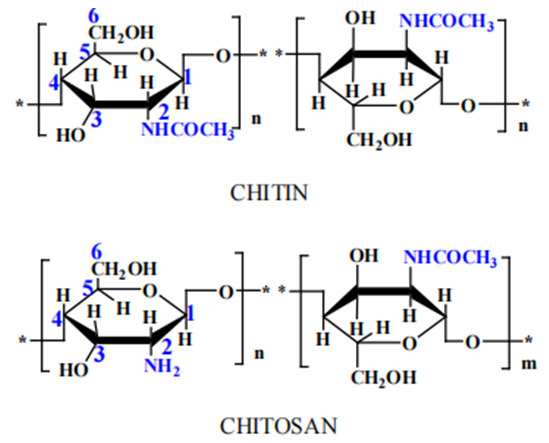
Surface modification of textile materials is considered as the best way to obtain modern textile treatment. Synthetic chemicals as coating material create an adverse impact on the environment. By using the natural properties of chitosan and egg white, the research work aimed to enhance fabric ...
Full-Text HTMLDownload Full-Text PDF
6972 Views 4416 Downloads
Synthesis and Composition of Tutton Double Salts: An Introductory Laboratory Project
Corissa P. McDonald, Claude H. Yoder
Journal of Laboratory Chemical Education, 2025, 13(1), pp. 16-24
DOI: 10.5923/j.jlce.20251301.02
Open Access
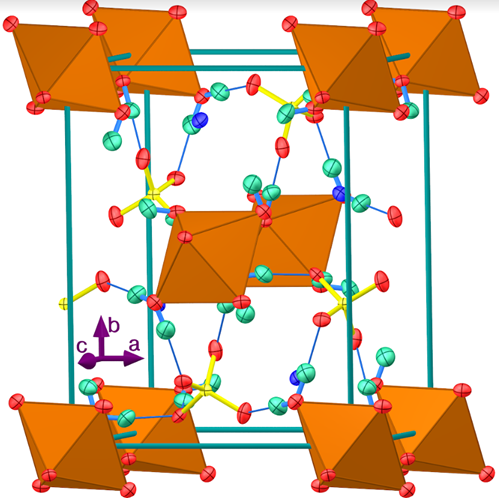
The synthesis and characterization of Tutton double salts is described as an ideal introduction to the synthesis and gravimetric analysis of soluble ionic compounds. The Tutton salts, with a formula of K
2M(SO
4)
2•6H
2O, where M is Mg, Zn, Cu, Ni, Co, and Fe, ...
Full-Text HTMLDownload Full-Text PDF
8027 Views 4775 Downloads